Product details
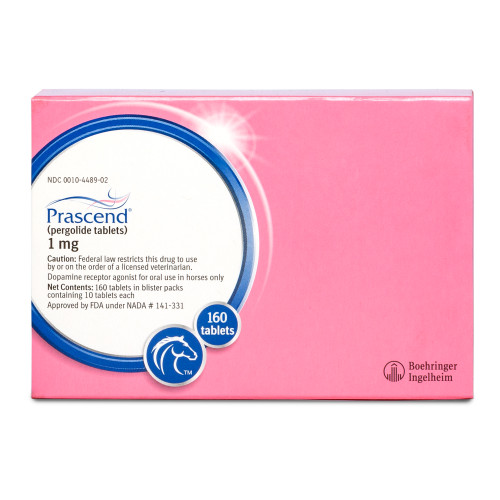
Zygolide (pergolide tablets) is indicated for the control of clinical signs associated with Pituitary Pars Intermedia Dysfunction (Equine Cushing's Disease) in horses.
-
The first FDA-approved generic peppermint-flavored pergolide tablet for the treatment of Pituitary Pars Intermedia Dysfunction (PPID) in horses
-
Peppermint flavor to support consistent daily use
-
360-degree, half-scored tablets for flexible, accurate dosing
-
Cost-effective treatment1
-
Do not crush due to the potential for increased human exposure
References:
1. Market data on file, Dechra Veterinary Products.
Product sourced directly from the manufacturer or their approved distributor. Guaranteed genuine product backed by the manufacturer.
For
Horses
Active ingredient(s)
Pergolide mesylate
Inactive ingredients
Complete inactive ingredients list not provided by manufacturer.
Brand name
Zygolide
Drug class
Dopamine receptor agonist
Product flavoring
Zygolide tablets are peppermint flavored.
Manufacturer
Dechra
Product strength & size
Zygolide contains 1 mg of pergolide in each tablet — packaged 10 tablets per blister. Please ensure the selected product and package size are appropriate for your horse, as recommended by your veterinarian.
For current availability, please refer to the product selection above.
Usage
For additional product information, please refer to the product label and the full Prescribing Information provided in the manufacturer’s package insert.
Zygolide tablets should be given orally as directed by your veterinarian. Please consult your veterinarian before making any changes or discontinuing the prescribed dosing schedule.
Manufacturer Dosage and Administration:
Zygolide should be administered orally at a starting dose of 2 mcg/kg once daily. Dosage may be adjusted to effect, not to exceed 4 mcg/kg daily.
The tablets are scored and the calculated dosage should be provided to the nearest one-half tablet increment.
|
Dosing Table
|
|---|
|
Body Weight
|
2 mcg/kg Dosage
|
4 mcg/kg Dosage
|
|---|
|
136 - 340 kg (300 - 749 lb)
|
0.5 tablet
|
1 tablet
|
|
341 - 567 kg (750 - 1,249 lb)
|
1 tablet
|
2 tablets
|
|
568 - 795 kg (1,250 - 1,749 lb)
|
1.5 tablets
|
3 tablets
|
|
796 - 1,022 kg (1,750 - 2,249 lb)
|
2 tablets
|
4 tablets
|
Missed doses
Administer the dose as soon as possible. If it is almost time for the next dose, skip the missed dose and continue with the regular schedule. Do not give more medication than prescribed or two doses at once.
If you have questions about your horse’s prescribed dosing schedule (e.g., timing, interval), please consult your veterinarian.
Storage
Store in a cool, dry place at a controlled room temperature at or below 25°C (77°F) with excursions up to 40°C (104°F). Keep in a secure location out of the reach of children and animals. Store this product separately away from human medicinal products and handle this product with care to avoid accidental ingestion.
Exposure to extreme temperatures, light, or moisture can affect the integrity and effectiveness of medications. Always store medications properly (e.g., away from the bathroom, kitchen sink, or other damp places).
Side effects
For complete product information, including potential side effects, please refer to the manufacturer's package insert.
As with all drugs, side effects may occur; the most common side effect of this medication is decreased appetite, which is typically mild. If severe, your veterinarian may recommend a temporary dose reduction.
Contact your veterinarian immediately if colic, laminitis, lameness, or fainting/collapse occurs.
This is not a comprehensive list of all possible side effects; for more information, please contact your veterinarian or refer to the manufacturer package insert. Notify your veterinarian if your horse experiences any of the effects described above, or if you notice any other side effects that are persistent or troublesome.
If you notice anything unusual, please consult your veterinarian.
Precautions
Before using this product, please review the product label and package insert for important safety information, including any relevant warnings and precautions.
For oral use in horses only. Do not use in horses intended for human consumption.
Not for use in humans. Zygolide should not be administered by persons who have had adverse reactions to ergotamine or other ergot derivatives.
Pregnant or lactating women should wear gloves when administering this product. It has been reported that pergolide tablets may cause eye irritation, an irritating smell, or headache when split or crushed. Zygolide tablets should not be crushed due to the potential for increased human exposure and care should be taken to minimize exposure when splitting tablets.
In case of accidental ingestion, seek medical advice immediately and show the package leaflet or the label to the physician.
Keep this and all medications in a secure location out of the reach of children and animals. Dogs have eaten pergolide tablets that were placed in food intended for horses or dropped during administration of the tablets to the horses. Adverse reactions may occur if animals other than horses ingest Zygolide tablets.
The use of Zygolide in breeding, pregnant, or lactating horses has not been evaluated. The effects of pergolide mesylate on breeding, pregnant, or lactating horses are not known; however, the pharmacologic action of pergolide mesylate suggests that it may interfere with reproductive functions such as lactation.
Do not administer this medication to horses with a known or suspected allergy/hypersensitivity to any ergot derivative, including pergolide mesylate, or any of its ingredients. Allergic reactions to medications may occur. Please inform Vetsource and your veterinarian if your horse has any known sensitivities or allergies, especially to medications and supplements.
If your horse displays symptoms of an allergic reaction, discontinue therapy and call your veterinarian immediately or seek emergency veterinary attention. Symptoms may include (but are not limited to): swollen lips, tongue, face, or airways; difficulty breathing; agitation; profuse salivation; and widespread hives or itching.
Drug and food interactions
Pergolide tablets are approximately 90% associated with plasma proteins. Use caution if administering Zygolide with other drugs that affect protein binding.
Dopamine antagonists, such as neuroleptics (phenothiazines, domperidone) or metoclopramide, ordinarily should not be administered concurrently with Zygolide (a dopamine agonist) since these agents may diminish the effectiveness of Zygolide.
*NOTE: This may not be a comprehensive list of all possible drug interactions. As with all medications, please consult your veterinarian if you have any questions or concerns prior to use. Contact your veterinarian if your horse experiences any unusual reactions when multiple medications and/or supplements are given together.
Please ensure your veterinarian is aware of all medications, supplements, and over-the-counter (OTC) products that your horse is currently using. Your veterinarian may prescribe multiple medications, even if a potential drug interaction may occur. In these instances, your veterinarian may adjust the dosages or monitor your horse more closely.
Adverse reactions
If you are concerned that your horse has experienced an adverse reaction to this medication, please contact the manufacturer Dechra Veterinary Products at 1-866-933-2472 or Vetsource Pet Owner Care at 877-738-4443.
Overdose
If you have any reason to suspect an overdose, call your doctor/veterinarian or the appropriate poison control resource immediately.
For humans:
The national toll-free Poison Help line, 1-800-222-1222, will connect you to your local poison center in case of emergency. This service is available nationwide and in most U.S. territories.
For animals:
The ASPCA Animal Poison Control Center is available 24 hours a day, 365 days a year at 888-426-4435.
Pet Poison Helpline® also provides a 24/7 animal poison control service at 855-764-7661.
*Please note: this information is for third-party services and is provided for convenience in case of potential poison-related emergencies. There may be consultation fee for these services.
Disclaimer
The content provided on this page is NOT medical advice.
All content, including the images and product description above, is intended for general informational purposes only and should not be considered a substitute for professional veterinary consultation, diagnosis, or treatment.
For additional information about this product and how it fits into your horse's individual treatment plan, please consult your veterinarian.
Last revised: 3/04/2026